Symeres has announced it has been awarded ISO/IEC 27001 certification, the internationally recognized standard for information security management systems (ISMS).
As scientific collaboration becomes increasingly digital and globally interconnected, amid rising cybersecurity threats and evolving AI-driven risks, biopharmaceutical companies are demanding more robust data protection protocols from outsourcing partners.
“Cybersecurity is no longer a back-office issue; it is central to the success of every client program,” said Gabriella Gentile, Chief Operating Officer at Symeres. “Our ISO 27001 certification gives clients, from biotech startups to large pharmaceutical companies, the assurance that their proprietary data, often the ‘crown jewels’ of development, is protected within a robust and continuously improving security framework.”
For many large pharmaceutical companies, information security assessments are now a standard part of onboarding. ISO 27001 certification allows Symeres to address the majority of these requirements upfront, significantly streamlining due diligence processes. By reducing administrative friction, project teams can focus more quickly on what matters most: advancing client programs and delivering scientific impact.
“This journey began two years ago as part of our continuous improvement culture,” added Mark Verhaar, Project Manager at Symeres. “We identified our highest information risk areas, developed risk-based policies, and created a formal, auditable system for managing access, backups, and incident response.”
“It’s not just about compliance,” he added. “It’s about building a culture of vigilance and continuous improvement, ensuring that our security practices evolve alongside the scientific and digital complexity of our clients’ programs.”
With ISO 27001 in place, Symeres is well-positioned to support the needs of both established pharmaceutical clients and smaller biotechs that may rely on its expertise to navigate complex data governance. The certification also supports broader industry compliance efforts, including the EU’s NIS2 Directive, which identifies healthcare organizations as vital operators.
Key elements of Symeres’s information security framework include:
- Formalised, audited policies for data access, backup, and breach response
- Continuous employee awareness programs to mitigate human error
- Controlled information sharing protocols with third-party partners and clients
- Ongoing internal audits and yearly external surveillance to maintain certification
Symeres is progressing with the rollout of ISO 27001 certification across its sites globally, with completion targeted before the end of the year. This phased implementation ensures consistent standards across the Group while maintaining operational continuity.
As cyber threats grow more sophisticated, maintaining high security standards will remain a priority.
Guillaume Jetten, Chief Executive Officer at Symeres, commented: “In today’s environment, ISO 27001 is a non-negotiable foundation for doing business with large pharmaceutical companies and innovative biotechs. Achieving this certification cements Symeres’ status as a leading global CDMOs operating with full transparency and accountability.”
Latest news

What are the top drug development trends for 2026? Exemplify CSO Paul O’Shea discusses what to watch

Symeres appoints Jurgen Berendsen as Chief Financial Officer

Symeres earns first SBTi approval as part of long-term sustainability journey

Symeres appoints Gabriella Gentile as Chief Operating Officer to drive transatlantic growth

Symeres announces acquisition of DGr Pharma

Symeres and Yoneda Labs use AI to optimize cross-coupling reactions

New production lines D1 and D2 at our Prague site

New NMR technology – meet “Maggie”

Symeres announces leadership evolution

Official opening of new Oncolines home, the Marie Curie Building at the Pivot Park in Oss, the Netherlands

New equipment at Exemplify

New 100 L vessels in Groningen

Organix: New facility

Innovative macrocycles as powerful Mcl-1 inhibitors

Oncolines unveils new state-of-the-art laboratories

Peter Molenveld assumes role as secretary of KNCV (Royal Netherlands Chemical Society)

Chiral chemistry: in the DNA of Symeres
π-facial selectivity in the Diels–Alder reaction of glucosamine-based chiral furans and maleimides

Symeres acquires Oncolines, further strengthening its drug discovery and biology capabilities

Symeres acquires Exemplify BioPharma, further strengthening its strategic foothold in the US

Expansion of solid-state center of excellence

Symeres acquires Massachusetts-based Organix Inc., adding lipids expertise and creating a strategic foothold in the US market
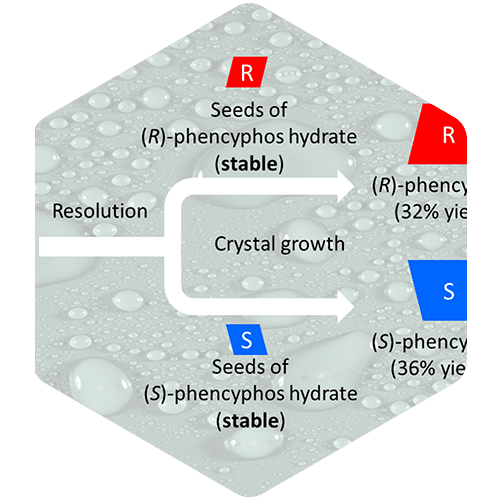
Expanding the toolbox: Resolving racemates

Symeres joins forces with Keensight Capital

Speak with our experts
Let’s discuss how Symeres can support the discovery and development of your next breakthrough