Symeres has been featured in a recent Speciality Chemicals article exploring how biotech companies are adapting to increasingly selective funding conditions, tighter timelines and growing pressure to demonstrate value earlier in development.
In the feature, Philip Payne, Chief Commercial Officer, and Dr Goran Verspui, Head of Drug Development Services at Symeres, discuss how modern biotech development models are evolving — and why integrated scientific expertise, connected workflows and faster decision-making are becoming increasingly critical for programme success.
The article examines how investor expectations are changing across the biotech sector, with funding increasingly focused on programmes capable of demonstrating strong data, reduced risk and clear development strategies earlier in the journey. As a result, biotech companies are placing greater emphasis on scientific integration, operational flexibility and technologies that accelerate high-quality decision-making.
The feature also explores:
- how advanced chemistry approaches and AI-enabled optimisation are improving development efficiency
- the importance of connected drug substance and drug product workflows
- why integrated scientific collaboration can help reduce delays and development risk
- how transatlantic partnerships support flexibility, resilience and global programme progression
- the growing strategic value of mid-sized CRDMOs in today’s biotech landscape
The article highlights how combining scientific depth, digital innovation and collaborative partnership models can help biotech companies navigate increasing development complexity while maintaining the speed needed to reach critical milestones.
Read the full feature: Navigating biotech’s new reality: Funding, flexibility & faster decision making
Latest news

Symeres appoints Henning Steinhagen as CEO to drive next phase of strategic growth
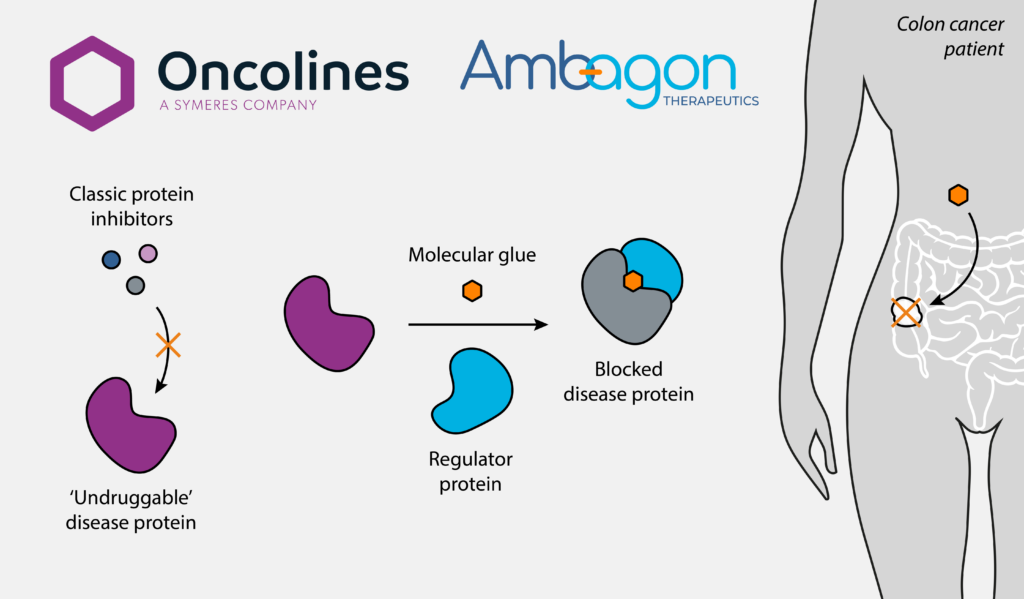
Symeres collaborates with Ambagon Therapeutics on molecular glue research in colorectal cancer

Symeres achieves ISO 27001 certification, reinforcing commitment to information security

What are the top drug development trends for 2026? Exemplify CSO Paul O’Shea discusses what to watch

Symeres appoints Jurgen Berendsen as Chief Financial Officer

Symeres earns first SBTi approval as part of long-term sustainability journey

Symeres appoints Gabriella Gentile as Chief Operating Officer to drive transatlantic growth

Symeres announces acquisition of DGr Pharma

Symeres and Yoneda Labs use AI to optimize cross-coupling reactions

New production lines D1 and D2 at our Prague site

New NMR technology – meet “Maggie”

Symeres announces leadership evolution

Official opening of new Oncolines home, the Marie Curie Building at the Pivot Park in Oss, the Netherlands

New equipment at Exemplify

New 100 L vessels in Groningen

Organix: New facility

Innovative macrocycles as powerful Mcl-1 inhibitors

Oncolines unveils new state-of-the-art laboratories

Peter Molenveld assumes role as secretary of KNCV (Royal Netherlands Chemical Society)

Chiral chemistry: in the DNA of Symeres
π-facial selectivity in the Diels–Alder reaction of glucosamine-based chiral furans and maleimides

Symeres acquires Oncolines, further strengthening its drug discovery and biology capabilities

Symeres acquires Exemplify BioPharma, further strengthening its strategic foothold in the US

Expansion of solid-state center of excellence

Symeres acquires Massachusetts-based Organix Inc., adding lipids expertise and creating a strategic foothold in the US market
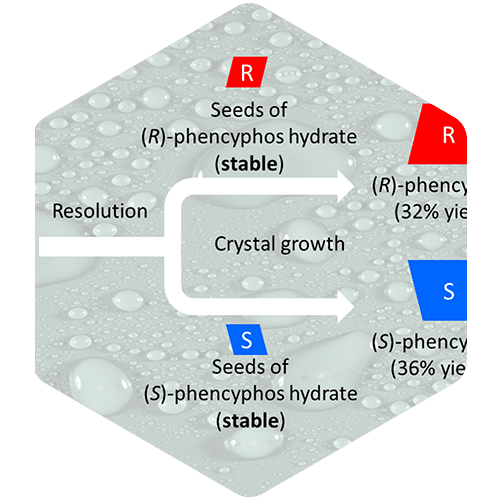
Expanding the toolbox: Resolving racemates

Symeres joins forces with Keensight Capital

Speak with our experts
Let’s discuss how Symeres can support the discovery and development of your next breakthrough