-
CDMO Red Flags You Can’t Ignore: Underestimating Technology Transfer Complexity
Transitioning a process from discovery scale to GMP manufacturing is almost never straightforward. What runs smoothly at 100 milligrams in the lab can behave very differently at the kilogram scale. Yet too many programs falter because the complexity of this transition is underestimated or treated as a simple “copy and paste” exercise.
-
O.N.E Symeres: A Practical Approach to Real-World Drug Development
No drug development program runs perfectly. Chemistry misbehaves, funding shifts, and timelines tighten. But what defines a reliable partner is how they respond. O.N.E Symeres is the framework we use to keep projects moving through uncertainty: openness, nimbleness, and expertise.
-
CDMO Red Flags You Can’t Ignore: Undefined or Shifting Project Scope
Selecting the right CDMO is one of the most important decisions in drug development. Yet even experienced biotechs and pharma companies can find themselves trapped in projects where the initial excitement gives way to frustration, and one of the most common culprits is a poorly defined scope.
-

Symeres Appoints Gabriella Gentile as Chief Operating Officer to Drive Transatlantic Growth
With more than 25 years of international experience in the pharmaceutical, medical device, and services sectors, Gabriella brings a proven track record in operational performance, lean methodologies, and global transformation programs.
-
Symeres Announces Acquisition of DGr Pharma
DGr Pharma specializes in preclinical and clinical regulatory strategy and consultancy for biotech and pharmaceutical partners working in early drug development. Its core services include chemical-pharmaceutical, non-clinical and clinical development planning, quality assurance, and regulatory submissions. DGr Pharma has deep expertise in small and large molecules including antibodies, ADCs, and oligonucleotides.
-
Insights into Drug Discovery and Development 2025
Here we interview our Director of Medicinal Chemistry, Anita Wegert, for her insights into drug discovery and development for 2025. This interview was conducted an interviewer from the Drug Discovery and Development Europe event and we were able to share our expertise. Curious how our insights can help your next project?
-
Symeres and Yoneda Labs Use AI to Optimize Cross-Coupling Reactions
San Francisco, May 29, 2025 – Yoneda Labs, a leader in computational tools for reaction optimization, has successfully collaborated with Symeres, a leading transatlantic, small molecule contract research and manufacturing organization. The aim was to optimize transition metal catalyzed cross-coupling reactions to improve reaction yield and efficiency utilizing Yoneda Labs’ cutting-edge software.
-
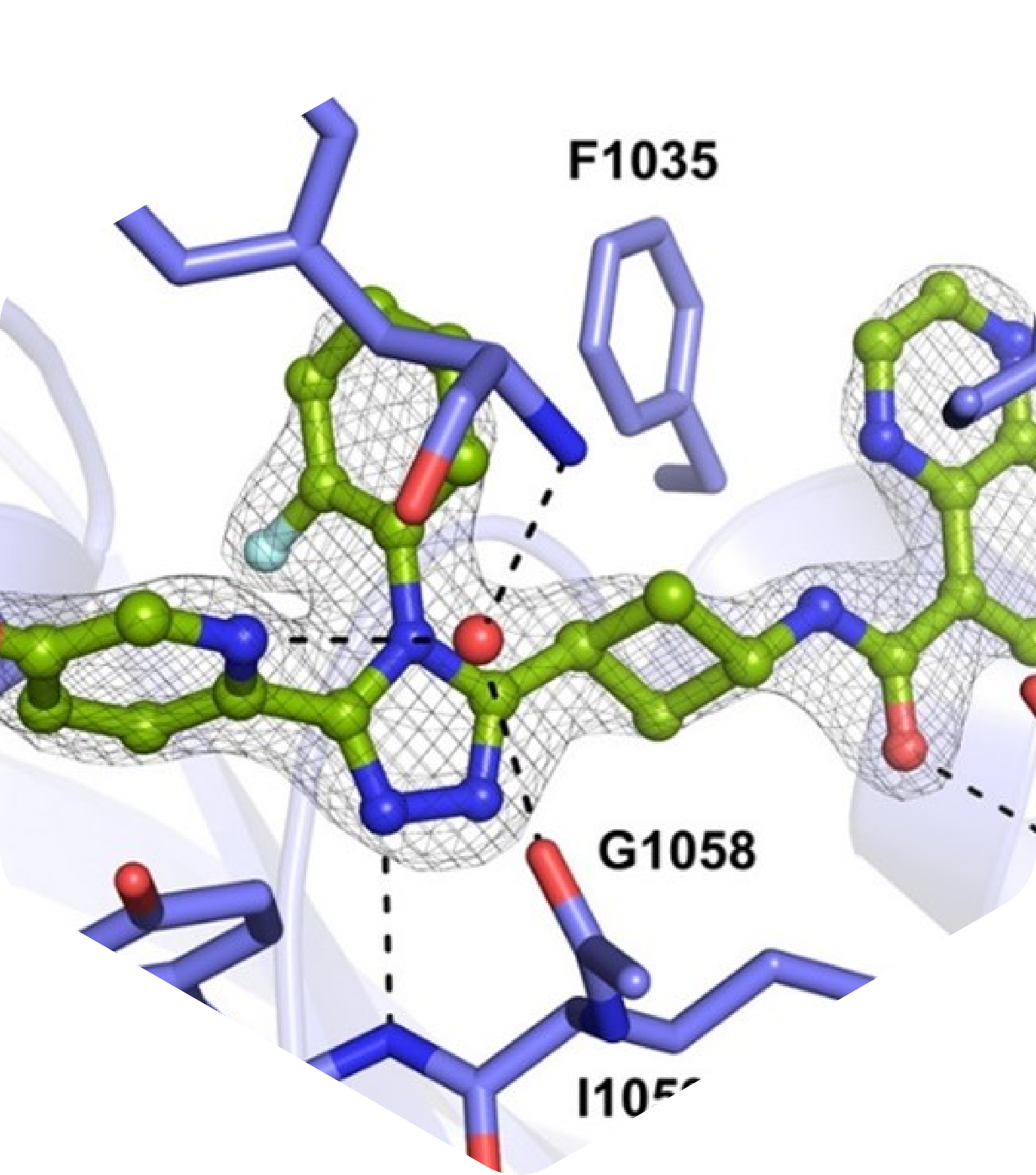
J. Med. Chem. Publication: Development of a 1,2,4-Triazole-Based Lead Tankyrase Inhibitor: Part II
In this follow-up article, co-published with Oslo University Hospital, we report further development and optimization of 1,2,3-triazole-based tankyrase inhibitors. Through X-ray crystallography supported SAR analysis, we synthesized a series of novel TNKS inhibitors with improved ADME/PK profiles, exhibiting antiproliferative efficacy in colorectal cancer cell lines. Click on the link below for a short summary of this publication.
-
Meet the Organix Director, Mario Gonzalez
We are pleased to share a conversation with Dr. Mario Gonzalez, a Director at Organix, as he reflects on his journey from Argentina to Massachusetts and provides valuable career insights in celebration of his 30 years with the company.
-
New Production Lines D1 and D2 at our Prague site!
The new production lines D1 and D2 were launched for the first time in May 2024 at the Prague site. The first batch of 11 kg of API was produced in the following weeks, and the tremendous efforts of the many people involved in planning the entire production line have finally paid off. Since the production line was launched, all equipment has been continuously utilized.
-
Reliable Tablet Formulation at Exemplify!
We at Symeres pride ourselves on a stellar track record in drug-substance development. For our clients that are looking to rapidly move through phase-appropriate clinical tablet formulation development, Exemplify is the perfect place to start for small-scale to large-scale pharma companies. Our cGMP suites are specially equipped to support everything from pre-tox formulation to first-in-human (FIH) trials and commercially ready formulation.
-
Peter Molenveld’s 25th Anniversary with Symeres
Recently, our colleague Peter Molenveld celebrated his 25th anniversary with Symeres. Peter participated in an interview where he shared about his time with Symeres and his growth here as a chemist to leadership within the organization, as well as about his role as secretary at the Royal Netherlands Chemical Society (KNCV).
Feel free to contact us!
Curious to know more about what can we do for you? Get in touch and let’s start a conversation.